|
Can two chemicals have the same chemical formula?
| Q: |
I saw on another site that the chemical formula for cocaine is
listed is C17H21NO4., but you have this formula listed on your datura page as the formula for scopolamine, isn't the datura chemistry page wrong? |
|
| A: |
Simple chemical formulas denoting the number of atoms in a molecule are not unique identifiers. There are a large number of chemicals which could all have the same empirical formula C17H21NO4.
What this formula means is simply that there are 17 Carbon atoms, 21 Hydrogens, 1 Nitrogen, and 4 Oxygens that make up this particular chemical. Those can be arranged in a large variety of ways, from straight lines, to what are called "benzene rings" , to other more complicated ring structures like the one that scopolamine has.
Scopolamine 3D Chemical Image
The right hand end of this image is a benzene ring, which is 6 carbon atoms attached in a 'flat' circle. It is perhaps interesting to note that both scopolamine and atropine have the same empirical chemical formula, but are distinct chemicals with different effects and
different structures. Take a look at the Datura Chemistry Page and look for the differences in the structure.
You can look at cocaine and scopolamine side by side
using Chem-Compare. You'll see that cocaine and scopolamine have a lot of similarities in general shape, size, and structure. But the two chemicals have profoundly different effects on the human nervous system. To compare them, start at the benzene ring on each of them and slowly work your way out into the rest of the molecule. You'll start to see differences very quickly.
I think that a fairly good example of the differences in structure for a very similar set of atoms can be seen
by comparing GBL and 1,4-butanediol. These molecules have the same number of carbon atoms and oxygen atoms, yet these are arranged differently. Although they do not have the same number of hydrogen atoms (1,4-butanediol has 10 hydrogens where GBL has 6), I think this can help give a sense of how knowing the empirical chemical formula does not necessarily tell you which chemical you have.
We drew another, more precise, example showing two chemicals side-by-side that are both very simple molecules, have identical empirical formulas (C4H10), yet have different structures and chemical properties. These chemicals, 2-methylpropane (also known as Isobutane) and n-butane look like:
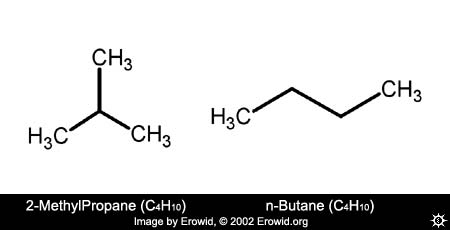
I hope that helps,
earth |
|
|
|
|
|
Categories:
[ Chemistry ]
|
|



