
#139 ORTHO-DOT
4,5-DIMETHOXY-2-METHYLTHIOAMPHETAMINE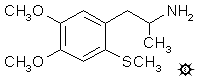
|
| [3D .mol structure] |
The finely pulverized 3,4-dimethoxybenzenesulfonyl chloride (33 g) was added to 900 mL of crushed ice in a 2 L round-bottomed flask equipped with a heating mantle and reflux condenser. There was then added 55 mL concentrated H2SO4 and, with vigorous mechanical stirring, there was added 50 g of zinc dust in small portions. This mixture was heated until a vigorous reaction ensued and refluxing was continued for 1.5 h. After cooling to room temperature and decantation from unreacted metallic zinc, the aqueous phase was extracted with 3x150 mL Et2O. The pooled extracts were washed once with saturated brine and the solvent was removed under vacuum. The residue was distilled to give 20.8 g of 3,4-dimethoxythiophenol boiling at 86-88 °C at 0.4 mm/Hg.
A solution of 10 g 3,4-dimethoxythiophenol in 50 mL absolute EtOH was protected from the air by an atmosphere of N2. There was added a solution of 5 g 85% KOH in 80 mL EtOH. This was followed by the addition of 6 mL methyl iodide, and the mixture was held at reflux for 30 min. This was poured into 200 mL H2O and extracted with 3x50 mL Et2O. The pooled extracts were washed once with aqueous sodium hydrosulfite, then the organic solvent was removed under vacuum. The residue was distilled to give 10.3 g of 3,4-dimethoxythioanisole with a bp of 94-95 °C at 0.4 mm/Hg. The product was a colorless oil that crystallized on standing. Its mp was 31-32 °C.
To a mixture of 15 g POCl3 and 14 g N-methylformanilide that had been warmed briefly on the steam bath there was added 8.2 g of 3,4-dimethoxythioanisole, the exothermic reaction was heated on the steam bath for an additional 20 min, and then poured into 200 mL H2O. Stirring was continued until the insolubles had become completely loose and granular. These were removed by filtration, washed with H2O, sucked as dry as possible, and then recrystallized from 100 mL boiling EtOH. The product, 4,5-dimethoxy-2-(methylthio)benzaldehyde, was an off-white solid, weighing 8.05 g and having a mp of 112-113 °C. Anal. (C10H12O3S) C,H.
A solution of 2.0 g 4,5-dimethoxy-2-(methylthio)benzaldehyde in 8 mL nitroethane was treated with 0.45 g anhydrous ammonium acetate and heated on the steam bath for 4.5 h. Removal of the excess solvent under vacuum gave a red residue which was dissolved in 5 mL boiling MeOH. There was the spontaneous formation of a crystalline product which was recrystallized from 25 mL boiling MeOH to give, after cooling, filtering and air drying, 1.85 g of 1-(4,5-dimethoxy-2-methylthiophenyl)-2-nitropropene as bright orange crystals with a mp of 104-105 °C. Anal. (C12H15NO4S) C,H,N.
A suspension of 1.3 g LAH in 50 mL anhydrous THF was placed under an inert atmosphere and stirred magnetically. When this had been brought to reflux conditions, there was added, dropwise, 1.65 g of 1-(4,5-dimethoxy-2-methylthiophenyl)-2-nitropropene in 20 mL THF. The reaction mixture was maintained at reflux for 18 h. After being brought back to room temperature, the excess hydride was destroyed by the addition of 1.3 mL H2O in 10 mL THF. There was then added 1.3 mL of 3N NaOH followed by an additional 3.9 mL H2O. The loose, inorganic salts were removed by filtration, and the filter cake washed with additional 20 mL THF. The combined filtrate and washes were stripped of solvent under vacuum yielding a light yellow oil as a residue. This was dissolved in 20 mL IPA, neutralized with 0.9 mL concentrated HCl, and diluted with 200 mL anhydrous Et2O. There was thus formed 1.20 g of 4,5-dimethoxy-2-methylthioamphetamine hydrochloride (ORTHO-DOT) as a pale yellow crystalline product. This melted at 218-219.5 °C, and recrystallization from EtOH yielded a white product and increased the mp to 222-223 °C with decomposition Anal. (C12H20ClNO2S) C,H,N.
DOSAGE: greater than 25 mg.
DURATION: unknown.
QUALITATIVE COMMENTS: (with 25 mg) Vague awareness, with the feeling of an impending something. Light food sat uncomfortably. By the late afternoon there was absolutely nothing. Threshold at best.
EXTENSIONS AND COMMENTARY: This material, ORTHO-DOT, can be looked at as the sulfur homologue of TMA-2 with the sulfur atom located in place of the oxygen at the 2-position of the molecule. At what level this compound might show activity is completely unknown, but wherever that might be, it is at a dosage greater than that for the PARA-DOT isomer, ALEPH-1 (or ALEPH), which was fully active at 10 milligrams (ALEPH can be looked at as TMA-2 with the sulfur atom located in place of the oxygen at the 4-position of the molecule). A lot of variations are easily makable based on this structure, but why bother? ALEPH is the much more appealing candidate for structural manipulation.
| [ |
[Main Index] | [Forward |

