Spice & Spin-offs
Prohibition's High-Tech Cannabis Substitutes
Jun 2009
Citation: Erowid E, Erowid F. "Spice & Spin-offs: Prohibition's High-Tech Cannabis Substitutes." Erowid Extracts. Jun 2009;16:12-16.

|
These products are one of the most interesting developments in grey-market recreational drugs since the "research chemical" phenomenon of the early 2000s. This new evolution in psychoactives is made possible by sophisticated knowledge of chemistry, pharmacology, scientific research, drug detection methods, marketing, and international drug laws. The trend involves the sale of novel, putatively legal, functional psychoactives packaged in such a way as to hide the identity of the active compounds from both governmental agencies and from other manufacturers, since candidly marketing a new recreational chemical would shorten the period before it is copied or banned.
Spice: The Template
When we first read about Spice in online forums in early 2007, we immediately suspected that this "herbal" concoction might be something more novel than merely another smoking blend claiming cannabis-like effects. Though we had little to go on other than informed speculation and a few first-hand reports, the described effects seemed too strong and too similar to cannabis to be the result of the listed ingredients, none of which was known for having any noteworthy psychoactivity.
We were unable to find a U.S. vendor that sold Spice, and multiple European suppliers told us that the manufacturer stipulated that it not be sold into the United States. (Later packaging specifically states "Not for sale in the USA".) We ordered a package from a British vendor in March 2007, and quickly verified our suspicions via bioassay; the effects were obviously the result of a THC-like compound. We have been following the Spice phenomenon ever since.The original small (3" x 3.75") foil packets feature a distinctive eye logo atop a stylized leaf and contain what appears to be a blend of different dried and ground plant materials. The package lists 13 herbs and spices: baybean, blue lotus, dwarf scullcap, indian warrior, lion's tail, maconha brava, marshmallow, pink lotus, red clover, rose, Siberian motherwort, vanilla, and honey.
...the described effects seemed too strong and too similar to cannabis to be the result of the listed ingredients...
Although the 2008/2009 packaging clearly states that it is "not for human consumption", a claim repeated by many of the online vendors who sell it, The Psyche Deli tellingly trademarked Spice in Britain both as incense and as an herbal smoking product/tobacco substitute.1
Spice: Effects
We interviewed a number of people who had smoked the original Spice and some Erowid crew also reported on their experiences. Everyone agreed with web forum postings that the Spice smoke is harsh compared to good quality cannabis or hash, with a pleasant vanilla aroma reminiscent of flavored tobaccos. Although one person described experiencing no significant effects from Spice, most reported that the effects were very similar to those of cannabis, and nothing like the "maybe I feel something, maybe I don't"-type effects of most herbal cannabis replacements. Spice smokers reported feeling "stoned" or a "cannabis-like high", yet also consistently reported that the effects were not quite the same as cannabis. While specific differences have not been clearly identified, many descriptions suggest that Spice's effects have a stronger physical component at a given level of mental high.Though some of the herbs listed on the Spice packaging are known to have mild psychoactive effects, it was obvious to the Erowid crew that none of the herbs by themselves could be responsible for the level of effects that were reported. The only listed herb with which we were not familiar was indian warrior, presumably Pedicularis densiflora, which we later confirmed does not have effects similar to Spice when smoked.
It seems that the effects of Spice are so similar to natural cannabis that even experienced smokers might not be able to reliably tell them apart outside of the differences in smoke quality and aroma. This represents a truly new feat in the world of quasi-legal psychoactive drugs, and one that apparently has made the producers of the material a lot of money.
| Brands & Varieties |
Albino Rhino Buds, Aroma, Bombay Blue, Caneff 5 star, Chillin XXX, D-Raw, Dark Matter, Dream, Everlast, Ex-ses (Platinum), Experience: Chill, Experience: Ignite, Experience: Red Ball, Fusion, Galaxy, Genie, Gorilla, Herb Dream, Ice Bud Extra Cold, Kronic, Krypto Buds, Magic, Mojo, Moon Rocks, Pep Spice, Sence, Smoke, Solar Flare, Space, Space Truckin', Spice, Spice Arctic Synergy, Spice Tropical Synergy, Spice Diamond, Spice Gold, Spice Gold Spirit, Spice Silver, Spicey XXX, SpiceWorld420, Spice99 (Ultra), Spike99, Smoke, Splice Platinum, Star Fire, Yucatan Fire, Zohai, Zohai SX.
|
Cannabinoid Combination?
The consistency with which Spice was described as having effects very similar to cannabis quickly led us (and others) to suspect that these products contained at least one unnamed synthetic or extracted psychoactive chemical. The most likely candidates seemed to be synthetic cannabinoids. We had heard rumblings for nearly a decade about the idea of selling novel synthetic cannabinoids on the grey market. These discussions included talk of intentionally crafting combinations of two (or more) synthetic cannabinoids, in order to achieve the most pleasant or desirable cannabis-like high possible. It has long been theorized that the combination of cannabinoids present in the plant produces a unique effect that is different from the effect caused by any of its pure chemicals in isolation, and some research has supported this theory.2 Many people who are prescribed Marinol (synthetic THC) report that they don't like Marinol's effects as much as those of cannabis itself. In cannabis-using subcultures, it is widely discussed that there are at least two distinct effects of cannabis: the sedating ("stoney" / body high) and the stimulating ("trippy" / mental high). Different strains of cannabis are said to have more of one quality or the other.
There are hundreds of uncommon synthetic cannabinoid receptor agonists with effects that are not well documented. As early as September 2006, Internet forum rumors claimed that Spice contained a synthetic cannabinoid called HU-210,3 a chemical 50-400 times more potent than THC.4 Some who had tried HU-210 argued it couldn't be that chemical because HU-210's effects last much longer than cannabis. One pharmacologist speculated that a combination of an extremely low level of HU-210 and another cannabinoid might effectively mimic cannabis, with a shorter duration than a higher dose of HU-210 alone.
Regardless of the exact details, it was not difficult to imagine that, in theory, a chemist or company would be able to add one or more similar chemicals to a complex blend of herbs, creating an apparently herbal product with effects similar to cannabis.
In the Lab
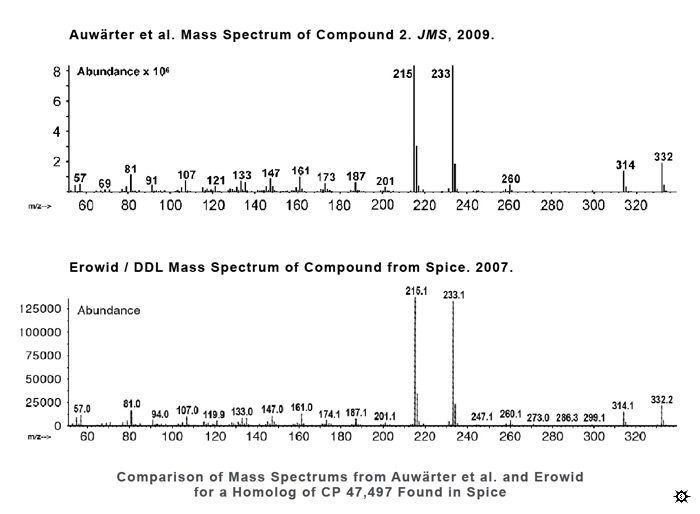
In April 2007, we sent a sample of basic Spice to Drug Detection Laboratories (DDL), the laboratory we work with for EcstasyData testing of street ecstasy tablets. We asked them to look for any identifiable chemicals, but also to specifically look for cannabinoids. DDL conducted GC/MS testing on the material, but their computerized matching system could not identify any known chemicals other than tocopherol (vitamin E, not a listed ingredient). The sample did not test positive for cannabinoids and the lab asserted that "no scheduled drugs [were] present".The mass spectrums, which could be thought of as the "chemical fingerprints" of the compound, included several major "peaks" that the lab was not able to identify. We asked other analytical chemists to look at the results, but had no luck discovering the identity of the mystery chemicals.
Difficulty of Detection
A common misconception about drug testing is that one can put an unidentified plant or mixture of chemicals in one end of an analytical machine, such as GC/MS or LC/MS, and the identities of its component substances will pop out the other end. When the analytical profile of a substance is not in a lab's database or if there are substances in a sample that have overlapping profiles, interpreting the results of GC/MS requires time and expertise. It is entirely possible for someone with knowledge of chemical analysis techniques to intentionally obscure the identity of the chemicals in a product. The most obvious way to do this would be to add a plant or chemical because it looks very similar in testing results to a chemical that the producers want to hide. When interpreting the results, many chemists would simply identify the presence of the plant ingredient without seeing that it obscured another chemical. It may be that the choices made about the ingredient profile in Spice were based, in part, on preventing easy detection.
|
Eureka: Identification
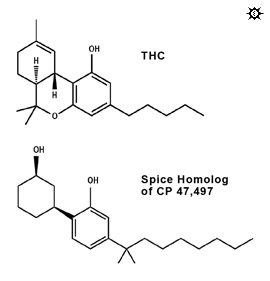
The same suspicions that we'd had about Spice led a number of organizations in various countries to do their own chemical testing. To date, a handful of synthetic chemicals have been identified in Spice-type products. In December 2008, the city of Frankfurt, Germany was the first to reveal definitive identification of a synthetic cannabinoid agonist in Spice products. They contracted with a specialized laboratory named THC Pharm to do an in-depth chemical analysis. THC Pharm found that Spice contained JWH-018, a cannabinoid agonist. In January 2009, U.S. Customs agents reported that seized Spice products contained HU-210, though theirs is the only lab to have found HU-210 and this finding has not yet been verified elsewhere.5In January 2009, the Journal of Mass Spectrometry published a letter by Auwärter, Dresen, et al. from the University of Freiburg that included mass spectrums (the "fingerprints") for several compounds found in Spice and related products. The authors identified the primary substance as an unnamed homolog of the cannabinoid agonist CP 47,497, with one more carbon atom on its "tail" than CP 47,497.6
We were excited to find that the mass spectrums of the compounds that were identified in the Auwärter paper as the primary cannabinoid agonists in Spice, Spice Gold, and Spice Diamond, nearly exactly matched the results of our 2007 analysis. The CP 47,497 homolog was clearly the same chemical that our laboratory analysis had been unable to identify in the Spice we purchased and had analyzed some 20 months before Auwärter et al. purchased the Spice they analyzed.5
Some open questions remain: Might extremely low doses of HU-210 or other chemicals be present in products where they have not yet been found? What variations in combinations or levels are currently being sold? Are there other analytical issues preventing detection in some instances? What chemicals will be discovered in future products of this type?
Popularity
In May 2009, a search on "Spice Gold" yielded 521,000 hits on Google and dozens of vendors selling related products on Ebay.com. With prices for a packet sometimes exceeding the local price of cannabis ($35 to $45 per 3-gram packet, with one Romanian web site listing packets for $115 USD), there is clearly substantial interest in these products.They have been popular in Britain, Germany, and Romania, and have been distribution in other European countries and the United States. While Erowid has been unable to find solid numbers on sales, we've received unconfirmed reports that many head shops were selling thousands of dollars worth of Spice-type products per day in 2008; and the distributor, The Psyche Deli, reportedly increased its assets by over $1,000,000 between 2006 and 2007.7
The success of Spice in 2007 and 2008 has lead to a proliferation of more than 40 copycats, some with blatant knock-off names such as "Spicey", "Pep Spice", "Spike 99", or "Splice Platinum". Several of these even feature variations on the Spice logo, such as a monster eye or an eye in a triangle.
Stories in mainstream news sources, including a notable article in the British Financial Times, as well as alternative publications like The Entheogen Review and forums such as Bluelight.ru, have publicly broadcast the existence of Spice. The DEA included a story about Spice products in its March 2009 issue of Microgram, and European governments have made moves to control these materials.
A Foxy Dilemma #
For Erowid, the most difficult aspect of the Spice explosion has been deciding how to handle information about these products before they were verified to contain novel synthetic additives. When we first learned of Spice, we didn't know whether its effects were due to a relatively safe set of herbs with a surprising synergy; a relatively safe and known chemical; a harmful chemical added by producers simply intent on making profits; or an unknown chemical with an unknown safety profile. The fine line between describing a new psychoactive and acting as an unintended promoter of a potentially harmful and/or scam product presented a familiar dilemma.In 2000, Erowid began publishing experience reports about the grey-market research chemical 5-MeO-DiPT ("Foxy"). By posting information about the substance before its use had spread to a large number of users, we were inadvertently advertising for a chemical few people had been exposed to. Erowid was also accused of accelerating law enforcement interest and media attention on this new drug. That experience has informed our choices around when and how to begin publishing information related to novel compounds.
|
We concluded that publishing about theoretical synthetic ingredients would likely increase sales and increase law enforcement attention. It is a strange bind, knowing that—due to the current state of prohibition—dispersing information that should be considered cautionary would likely have the opposite effect.8
Safe as Weed?
Because Spice products are used as cannabis replacements, their safety must be compared with that of smoked cannabis. Unlike Ecstasy alternatives, which might offer more benign safety profiles than MDMA (lower risk of death or less neurotoxicity), Spice-type products are up against a mountain of historical and scientific evidence that establishes the safety of cannabis.Large epidemiological studies and experimental data have shown cannabis smoking does not involve many acute risks other than those of general inebriation.9 Even extended, heavy cannabis use does not appear to cause brain damage,10 nor substantial increases in risks of lung cancer11 or heart disease in healthy users;12 and it causes only moderate increases in respiratory illnesses.13 Further, THC has been shown to have anti-cancer properties in a number of experiments14,15 and has even been shown to be neuroprotective.10
Cannabis is, however, well known for causing some people to experience paranoid ideation and feelings of anxiety. The possibility of triggering psychosis is perhaps the top cannabis-related health issue receiving research attention in the last decade. Some psychoactive cannabinoid agonists other than THC might not cause paranoia effects or might be less likely to trigger latent psychoses, though there is little evidence that Spice products have either of those benefits.
Because Spice products are used as cannabis replacements, their safety must be compared with that of smoked cannabis.
There is an unmet public health need for government-sponsored testing of black- and grey-market recreational drugs. The producers of those products will never do adequate safety testing. Given that this is a persistent, long-term problem, public health organizations must step up to fund the development and administration of a standardized battery of tests for novel compounds. Such tests would ideally look for carcinogenicity, cardiovascular risks, and interactions with common recreational and medical drugs, and would also assess whether new compounds present other mental and physical health issues.
Hazards of Prohibition
Prohibition of widely used recreational drugs creates profitable markets for novel psychoactives. The unwillingness of governments around the world to authorize new recreational drugs means that such products will necessarily be unregulated. The UNODC estimates that there are over 150 million current cannabis users in the world, virtually all of whom use it illegally,19 suggesting that the market for effective cannabis replacements is hundreds of millions of dollars per year.Drug prohibition creates a pressure to develop new substances that are active at low doses, because they are easier to transport and more difficult to detect. Prohibition also drives illicit drug manufacturers to lie about the ingredients of newly designed products.
Incentives to disguise and misrepresent the contents of products arise not only from the threat of criminal penalty, but also from the realities of an unregulated marketplace. Producers who invest time and money in the development of new grey-market products must hide the details of their product from competitors (and thus from the public), in order to maximize their profit. Without the protections afforded by patents and governmental approvals, cheap knock offs hit the market as soon as the nature of Spice came to light.

|
The modern industrial product cycle can be extremely rapid, churning out new packaging and products in a matter of weeks. Manufacturers can not only vary the chemical profile of different product varieties, but also vary what chemicals are included in a product over time, shifting from one to another to stay ahead of the law and their competitors. Imagine the difficulty for customs agents trying to block importation of dozens or hundreds of professional-looking products whose packaging, marketing copy, and listed ingredients change seasonally.
A Taste of the Future
Spice is a fascinating test case for unusually potent drugs of the future. The Spice story is part sci-fi, part Prohibition-style bootlegging, and part crass commercial venture. This new generation of recreational "research chemical" cannabinoids and cannabinomimetics are in the first wave of high-tech crypto drugs, more of which may be just around the corner.
The evolutionary pressures of the last century have created a climate where new potent drugs are not only obscured, but obfuscated using specialized technical knowledge that even expert laboratories have difficulty sorting through. The Spice phenomenon gives us a glimpse of the complexities to be faced in the future when providing harm-reduction and health information for an ever-shifting, ever-expanding profusion of psychoactive drugs, drug combinations, and technologies.[See also: Spice Experience Report Excerpts]
References #
- UK Intellectual Property Office. Trademark 2477963A. 2008.
- Morgan CJ, Curran HV. "Effects of Cannabidiol on Schizophrenia-Like Symptoms in People Who Use Cannabis". Br J Psychiatry. 2008;192(4):306-7.
- Shamantra. "Spice Contains HU 210?". EveryoneDoesIt.com. Accessed May 23, 2007. Thread 74135.
- Devane WA, Breuer A, Sheskin T, et al. "A Novel Probe for the Cannabinoid Receptor". J Med Chem. 1992;35(11):2065-9.
- Auwärter V. Personal communication. Apr 2009.
- Auwärter V, Dresen S, Weinmann W, et al. "'Spice' and Other Herbal Blends: Harmless Incense or Cannabinoid Designer Drugs?". J Mass Spectrom. Feb 2, 2009;44(5):832-7.
- Jack A. "The Story of Spice". Financial Times. Feb 13, 2009.
- Brecher EM, Consumer Reports Eds. "How to Launch a Nationwide Drug Menace." The Consumers Union Report on Licit and Illicit Drugs. Part VI - Inhalants, solvents and glue-sniffing, Chapter 44. 1972.
- Sidney S. "Comparing Cannabis with Tobacco—again". BMJ. 2003;327(7416):635-6.
- Iversen L. "Cannabis and the Brain". Brain. 2003;126(Pt 6):1252-70.
- Hashibe M, Morgenstern H, Cui Y, et al. "Marijuana Use and the Risk of Lung and Upper Aerodigestive Tract Cancers". Cancer Epidemiol Biomarkers Prev. 2006;15(10):1829-34.
- Sidney S. "Cardiovascular Consequences of Marijuana Use". J Clin Pharmacol. 2002;42(11 Suppl):64S-70S.
- Tetrault JM, Crothers K, Moore BA, et al. "Effects of Marijuana Smoking on Pulmonary Function and Respiratory Complications: A Systematic Review". Arch Intern Med. 2007;167(3):221-8.
- Salazar M, Carracedo A, Salanueva IJ. "Cannabinoid Action Induces Autophagy-Mediated Cell Death Through Stimulation of ER Stress in Human Glioma Cells". J Clin Invest. 2009;119(5):1359-72.
- Guzman M. "Cannabinoids: Potential Anticancer Agents". Nat Rev Cancer. 2003;3(10):745-55.
- Anonymous. Personal Communication. 2009.
- Sand N. Personal Communication. 2009.
- Herkenham M, Lynn AB, Little MD, et al. "Cannabinoid Receptor Localization in Brain". Proc Natl Acad Sci. 1990;87(5):1932-6.
- UNODC. World Drug Report 2008. 2008.
- Boyer EW, Kearney S, Shannon MW, et al. "Poisoning from a Dietary Supplement Administered During Hospitalization". Pediatrics. 2002;109(3):E49.
- Gryniewicz CM, Reepmeyer JC, Kauffman JF, et al. "Detection of Undeclared Erectile Dysfunction Drugs and Analogues in Dietary Supplements by Ion Mobility Spectrometry". J Pharm Biomed Anal. 2009;49(3):601-6.

