
#170 5-TOET
4-ETHYL-2-METHOXY-5-METHYLTHIOAMPHETAMINE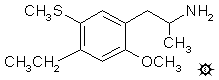
|
| [3D .mol structure] |
To a solution of 9.7 g 3-ethyl-4-(methylthio)phenol in 50 mL MeOH there was added a solution of 4.6 g 85% KOH in 50 mL hot MeOH. There was then added 5.4 mL methyl iodide and the mixture was held at reflux on the steam bath for 18 h. Removal of the solvent under vacuum gave a residue that was poured into 1 L H2O and made strongly basic by the addition of 5% NaOH. This was extracted with 3x75 mL CH2Cl2, and the extracts were pooled and the solvent removed under vacuum. There remained 11.0 g of an almost white oil with a startling apple smell. This oil was distilled at 78-88 °C at 0.3 mm/Hg to give 7.9 g 3-ethyl-4-(methylthio)anisole as a white oil. Anal. (C10H14OS) C,H.
A mixture of 7.8 g POCl3 and 6.9 g N-methylformanilide was heated on the steam bath for a few min, until there was the development of a deep claret color. This was added to 7.7 g 3-ethyl-4-(methylthio)anisole and the mixture was heated on the steam bath for 2 h. This was poured into 400 mL H2O and stirred overnight, which produced an oily phase with no signs of crystals. The entire reaction mixture was extracted with 3x75 mL CH2Cl2, and the pooled extracts washed with H2O. Removal of the solvent under vacuum gave 9.2 g of a residue. This was suspended in 25 mL hexane, and after 1 h standing, the overhead clear solution was decanted from the settled sludge. This hexane solution was stripped of solvent under vacuum, giving 7.7 g of an oil that by TLC was a mixture of starting ether and desired aldehyde. This was distilled at 0.25 mm/Hg to give three fractions, the first boiling at 75-100 °C (2.7 g) and the second at 100-115 °C (2.6 g). These were largely starting ether and aldehyde, and were chemically processed below. A third fraction, boiling at 120-140 °C, solidified in the receiver, weighed 1.6 g, and was largely the desired aldehyde. Cuts #1 and #2 (5.3 g of what was mostly recovered aldehyde) were resubmitted to the Vilsmeier reaction. A mixture of 5.4 g POCl3 and 4.7 g N-methylformanilide was heated on the steam bath until it became claret-colored. The recovered aldehyde was added, and the mixture was heated overnight on the steam bath. This was poured into 500 mL H2O. The heavy tar that was knocked out was extracted with 3x75 mL CH2Cl2, and the solvent was removed from the pooled extracts under vacuum. Some 5.8 g of residue was obtained, and this was heated to 120 °C at 0.2 mm/Hg to remove all materials lower boiling than the desired aldehyde. The very dark pot was extracted with 3x50 mL boiling hexane, and removal of the solvent from these pooled extracts under vacuum gave 0.9 g of a yellow oil. This was distilled at 0.2 mm/Hg to give a fraction boiling at 130-140 °C which spontaneously crystallized. This pressed on a porous plate gave almost white crystals with a mp of 55-57 °C. Recrystallization from 0.3 mL cyclohexane provided 0.3 g of 4-ethyl-2-methoxy-5-(methylthio)benzaldehyde with a mp of 57-58 °C. The total yield was 1.9 g. Anal. (C11H14O2S) C,H.
To a solution of 1.2 g 4-ethyl-2-methoxy-5-(methylthio)benzaldehyde in 25 mL nitroethane there was added 0.25 g anhydrous ammonium acetate and the mixture was heated on the steam bath. The initial color was green, but this quickly changed to the more usual yellow which darkened as the reaction mixture was heated. After 1.5 h heating, the excess solvent/reagent was removed in vacuo. The yellow residue was dissolved in 10 mL hot MeOH and allowed to stand in the refrigerator overnight. There was an orange oil layer formed underneath the MeOH. A small sample of this was scratched externally with dry ice, and seed was obtained. The orange oil layer slowly set to crystals which, after a few h, were removed by filtration to give 1.3 g of a slightly sticky orange solid with a mp of 43-45 °C. This was recrystallized from 8 mL boiling MeOH to give, after cooling, filtering, and air drying to constant weight, 1.1 g of 1-(4-ethyl-2-methoxy-5-methylthiophenyl)-2-nitropropene as electrostatic yellow crystals melting at 59-60 °C. Anal. (C13H17NO3S) C,H.
A solution of 1.0 g LAH in 25 mL tetrahydrofuran was cooled, under He, to 0 °C with an external ice bath. With good stirring there was added 0.6 mL 100% H2SO4 dropwise, to minimize charring. This was followed by the addition of 1.1 g of 1-(4-ethyl-2-methoxy-5-methylthio)-2-nitropropene in a small amount of THF. After 10 min further stirring, it was brought up to room temperature and allowed to stand for several days. The excess hydride was destroyed by the cautious addition of IPA followed by sufficient 15% NaOH to give a white granular character to the aluminum oxide, and to assure that the reaction mixture was basic. This was filtered, and the filter cake washed first with THF and then with IPA. The filtrate and washings were pooled and stripped of solvent under vacuum providing a pale amber residue. This was dissolved in 50 mL of dilute H2SO4 and washed with 2x50 mL CH2Cl2. The aqueous phase was made basic with 5% NaOH, and extracted wit 2x50 mL CH2Cl2. These extracts were pooled, stripped under vacuum, and distilled at 0.15 mm/Hg. The fraction with a bp of 102-128 °C weighed 0.4 g and was a colorless liquid. This was dissolved in a small amount of IPA, neutralized with concentrated HCl and diluted with anhydrous Et2O to provide the 4-ethyl-2-methoxy-5-methylthioamphetamine hydrochloride (5-TOET) which weighed 0.6 g and melted at 146-147 °C. Anal. (C13H22ClNOS) C,H.
DOSAGE: 12 - 25 mg.
DURATION: 8 - 24 h.
QUALITATIVE COMMENTS: (with 8 mg) After my totally freaky experience on the very closely related compound in this series, 5-TOM, I intended to approach this with some caution. Three milligrams was without effects, so I tried eight milligrams. I was a little light-headed, and saw sort of a brightness around trees against the blue sky. Noticed movement on couch in living room, and there was some activity in the curtains, almost 2C-B like. In the evening writing was still difficult, and there was eye dilation but minimal nystagmus. My sleep was fitful, but certainly there was no hint of the 5-TOM storm.
(with 18 mg) This was too much. There was an exhausting visual hallucinatory tinsel, continuous movement, and there was no escape. It popped into an LSD-like thing, strong, restless, constantly changing, with too much input. I had to take a Miltown to calm down enough for an attempt at sleep. In the morning, a day later, I was still 1.5 + and tired of it. It was the next day after that before I was completely clear.
(with 20 mg) This has the makings of a superb, extraordinary material. I didn't get to a full plus two, maybe something around a plus one and three quarters. The eyes-closed fantasy was exceptional, with new dimensions. The nature of the fantasy, the feeling that one had about the fantasy figures and landscapes, was the essence of joy, beauty, lovingness, serenity. A glimpse of what true heaven is supposed to feel like. Or maybe a button in the brain was pushed which has not been pushed by previous chemicals. Insight? Don't know yet. I was able to function without difficulty with eyes closed or open. Erotic absolutely exquisite. In fact, the entire experience was exquisite. Next day, same sense of serene, quiet joy/beauty persisted for most of the day. A true healing potential. Onwards and upwards. This one could be extraordinary.
(with 30 mg) Tried to focus on cosmic questions, and succeeded. Very little fantasy images for the first 2-3 hours. After that, lovely interacting, music okay but not vital. On this compound the Brahms Concerto #1 gave vivid 'memory' impressions of house and vegetable garden, like a primitive painting. Tremendous nostalgia for a place I've never seen.
EXTENSIONS AND COMMENTARY: With the extraordinary experience that had been observed with one person with 5-TOM, this ethyl homologue was not only run up with special caution, but that individual ran his own personal titration. And he proved to be perhaps twice as sensitive to 5-TOET than any of the other subjects. An approach to what might just be some unusual metabolic idiosyncrasy on the part of his liver, is discussed in the recipe for TOMSO.
The initials of TOET progressed quite logically from TOM, in an exact parallel with the relationship between the corresponding sulfur-free analogues, where the ethyl compound is DOET and the methyl counterpart is DOM. "T" for "thio" which is the chemical nomenclature term for the replacement of an oxygen atom with a sulfur atom. And, as has been discussed in the text of this volume, the peculiarities of pronunciation in this series are interesting, to say the least. TOM is no problem. But TOET could have any of several pronunciations such as "Two-it", or "Tow-it", or "Too-wet", but somehow the one syllable term "Twat" became regularly used, and the family was generally referred to as the "Toms and Twats." The almost-obscene meaning of the latter was progressively forgotten with usage, and has led to some raised eyebrows at occasional seminars when these compounds are discussed. And not only at seminars. Once at the between-acts intermission at the Berkeley Repertory Theater, the topic came up and the phrase was used. There was a stunned silence about us within the circle of hearing, and we seemed to have been given a little extra room immediately thereafter.
As with the other members of the TOM's and TOET's, the phenethylamine homologue of 5-TOET was synthesized, but had never been started in human evaluation. The aldehyde from above, 4-ethyl-2-methoxy-5-(methylthio)benzaldehyde, was condensed with nitroethane (as reagent and as solvent) and with ammonium acetate as catalyst to give the nitrostyrene as spectacular canary-yellow electrostatic crystals with a mp of 91-92 °C. Anal. (C12H15NO3S) C,H. This was reduced with aluminum hydride (from cold THF-dissolved lithium aluminum hydride and 100% sulfuric acid) to the phenethylamine 4-ethyl-2-methoxy-5-methylthiophenethylamine (2C-5-TOET) which, when totally freed from water of hydration by drying at 100 °C under a hard vacuum, had a mp of 216-217 °C. Anal. (C12H20ClNOS) C,H.
| [ |
[Main Index] | [Forward |

