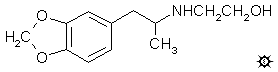
#107 MDHOET
HYDROXYETHYL-MDA; 3,4-METHYLENEDIOXY-N-(2-HYDROXYETHYL)AMPHETAMINE
SYNTHESIS: To a well stirred solution of 25 g ethanolamine
hydrochloride in 75 mL MeOH there was added 4.45 g of
3,4-methylenedioxyphenylacetone (see under MDMA for its preparation)
followed by 1.1 g sodium cyanoborohydride. Concentrated HCl in MeOH
was added as required, over the next few days, to maintain the pH at
about 6 as determined with external, dampened universal pH paper. The
reaction mixture was added to 300 mL H2O and made strongly acidic with
an excess of HCl. After washing with 3x100 mL CH2Cl2 the aqueous
phase was made basic with 25% NaOH, and extracted with 4x100 mL
CH2Cl2. Removal of the solvent under vacuum yielded 3.5 g of a
viscous off-white oil that was distilled at 160 °C at 1.3 mm/Hg to
give 2.0 g of a white viscous oil. The pot residue remained fluid,
but was discarded. This distillate was dissolved in 8.0 mL IPA to
give, eventually, a clear solution. This was neutralized with
concentrated HCl and diluted with 100 mL anhydrous Et2O. The loose
white crystals of 3,4-methylenedioxy-N-(2-hydroxy-ethyl)amphetamine
hydrochloride (MDHOET) that formed were removed by filtration, washed
with Et2O, and air dried. These weighed 2.3 g, and had a mp of
147-148 °C. Anal. (C12H18ClNO3) N.
DOSAGE: greater than 50 mg.
DURATION: unknown.
EXTENSIONS AND COMMENTARY: Most compounds with bare, exposed polar
groups like hydroxyls are not centrally active, as they simply do not
have any way of getting into the brain. MDHOET is certainly not very
active, if it is active at all.
There was one report that at very high doses some central effects were
indeed observed. With quantities in the several hundreds of
milligrams a picture emerged of changes in perceived color and depth
perception, but without euphoria. It was said to resemble a mild dose
of ketamine. This is an interesting comment, in that ketamine has
found its major medical use as an anesthetic, and MDHOET is among the
most effective of all the N-substituted MDA derivatives assayed in
several animal analgesia models.