
#88 HOT-7
2,5-DIMETHOXY-N-HYDROXY-4-(n)-PROPYLTHIOPHENETHYLAMINE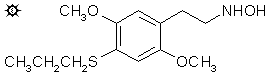
|
| [3D .mol structure] |
DOSAGE: 15 - 25 mg.
DURATION: 6 - 8 h.
QUALITATIVE COMMENTS: (with 15 mg) I am lightheaded, and maybe a little tipsy. I am well centered, but I don't want to go outside and meet people. Shades of alcohol woozy. The effects were going already by the fifth hour and were gone by the seventh hour. I would call it smoothly stoning.
(with 22 mg) The transition into the effects was a bit difficult, with a faint awareness in the tummy. But by the second hour it was quite psychedelic, and the body was not thought of again, except in terms of sexual fooling around. Very rich in eyes-closed imagery, and very good for interpretive and conceptual thinking. But the eyes-open visuals were not as much as they might have been. At the seventh hour, drifted into an easy sleep.
(with 22 mg) The experience was very positive, but at each turn there seemed to be a bit of sadness. Was it a complete plus three experience? Not quite. But it didn't miss by much. The erotic explorations somehow just failed to knit by the thinnest of margins. It was a truly almost-magnificent experience.
EXTENSIONS AND COMMENTARY: There is a working hypothesis that has been growing in substance over the last few years in this strange and marvelous area of psychedelic drugs. It all was an outgrowth of the rather remarkable coincidence that I had mentioned in the discussion that followed MDOH. There, an assay of what was thought to be MDOH gave a measure of activity that was substantially identical to MDA, and it was later found out that the material had decomposed to form MDA. So, MDA was in essence rediscovered. But when the true, valid, and undecomposed sample of MDOH was actually in hand, and assayed in its own rights, it was found to have a potency that really was the same as MDA. So, the working hypothesis goes something like this:
AN N-HYDROXY AMINE HAS APPROXIMATELY THE SAME POTENCY AND THE SAME ACTION AS ITS N-HYDROGEN COUNTERPART.
Maybe the N-hydroxy compound reduces to the N-H material in the body, and the latter is the intrinsically active agent. Maybe the N-H material oxidizes to the N-hydroxy material in the body, and the latter is the intrinsically active agent. Either direction is reasonable, and there is precedent for each. The equivalence of MDA and MDOH was the first suggestion of this. And I have made a number of NH vs. NOH challenges of this hypothesis. The interesting 2C-T-X series has provided a number of amines that are amenable to N-hydroxylation, and this is the first of them. And, after all, if you put a hydroxy (HO) group on a thio material (T), you have a HOT compound.
So, as far as nomenclature is concerned, the family of N-hydroxy analogues of N-H amines is known as the HOT family.
How does HOT-7 compare with 2C-T-7? They are almost identical. The same range of dose (centering on 20 milligrams) and if anything, perhaps slightly less long lived. Lets try some other N-hydroxys!
| [ |
[Main Index] | [Forward |

