
#67 DOI
2,5-DIMETHOXY-4-IODOAMPHETAMINE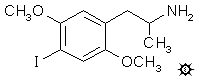
|
| [3D .mol structure] |
To a solution of 2.0 g N-(1-(2,5-dimethoxyphenyl)-2-propyl)phthalimide in 15 mL warm acetic acid which was being vigorously stirred, there was added a solution of 1.2 g iodine monochloride in 3 mL acetic acid. This was stirred for 2 h at about 40 °C during which time there was a definite lightening of color, but no solids formed. The reaction mixture was poured into 600 mL H2O which produced a reddish glob floating in a yellow-orange opaque aqueous phase. The glob was physically removed, dissolved in 30 mL boiling MeOH which, on cooling in an ice bath, deposited off-white crystals. These were removed by filtration, washed with MeOH, and air dried to give 1.5 g of N-[1-(2,5-dimethoxy-4-iodophenyl)-2-propyl]phthalimide as fine white crystals with a slight purple cast. The mp was 103-105.5 °C and the mixed mp with the starting non-iodinated phthalimide (mp 105-106 °C) was depressed (85-98 °C). Extraction of the aqueous phase, after alkalinification, provided an additional 0.15 g product. Anal. (C19H18NO4) C,H,N.
A solution of 0.75 g N-(1-(2,5-dimethoxy-4-iodophenyl)-2-propyl)phthalimide in 10 mL EtOH was treated with 0.3 mL of hydrazine hydrate, and the clear solution was held at reflux on the steam bath overnight. After cooling, there was a crystallization of 1,4-dihydroxyphthalizine that started as small beads but finally became extensive and quite curdy. These solids were removed by filtration and had a mp of about 340 °C (reference samples melted over a five to ten degree range in the area of 335-350 °C). The filtrate was dissolved in 100 mL CH2Cl2 and extracted with 2x150 mL 0.1 N HCl. The aqueous extracts were washed once with CH2Cl2, made basic with 5% NaOH, and extracted with 3x100 mL CH2Cl2. Removal of the solvent under vacuum gave 0.5 g of a colorless oil which was dissolved in 300 mL anhydrous Et2O and saturated with anhydrous HCl gas. There was obtained, after filtration, and air drying, 0.35 g of 2,5-dimethoxy-4-iodoamphetamine hydrochloride (DOI) as white crystals that melted at 200.5-201.5 °C. This value did not improved with recrystallization. Anal. (C11H17ClINO2) C,H,N.
DOSAGE: 1.5 - 3.0 mg.
DURATION: 16 - 30 h.
QUALITATIVE COMMENTS: (with 0.6 mg) There was a nice spacey light-headedness for a few hours, and time seemed to move quite slowly. Then a generic sadness came over me, as I reminisced about earlier days (recalling pleasures now gone) and wondered if I would be allowed to be here on the Farm when I am old and not important. There is so much to be done, and I cannot do it all, and no one else cares. My mood became present-day and healthy by about the seventh hour.
(with 1.6 mg) The general nature of the experience was depressing, with a sad view of life. There was no way I could connect with my emotions. Even my sadness was vague. At about the ninth hour I decided that enough was enough, and this strangely disappointing about-plus-two was aborted with 125 micrograms of LSD. The emotions became present and living within a half hour. I was greatly relieved. The erotic was not a mechanical attempt but a deeply involved feeling with an archetype of orgasm easily available. It was shaped like a flower, richly colored, with an unusual "S" shape to it. This was a lovely end to a difficult day.
(with 3.0 mg) This is a clear, clean psychedelic. The eyes-closed imagery is excellent, with clearly delineated patterns, pictures, and colors. Perfect for an artist, and next time I'll devote some time to painting. Total ease for the body, but no help for my smoking problem. I still want to smoke. And at sixteen hours into this I am still at 1.5+ but I'll try to go to bed anyway, and sleep.
(with 3.5 mg) I was at a full crashing +++ for about three or four hours. There was none of the LSD sparkle, but there were moments of `light-headedness' where one could move sideways with reality. I could leave where I was right over there, and come over here and get a strange but authentic view of where the `there' was that I had left. It would be out-of-body, except that the body came over here with me rather than staying there. This doesn't make sense now, but it sure did then. There was no trace of body impact, and I slept late that evening, but with some guardedness due to the intense imagery. This was no more intense than with 3.0 milligrams, but it was a little bit more to the unreal side.
(with 1.0 mg of the "R" isomer) There was a clear ++ from the second to the eighth hour, but somehow there was not quite the elegance or the push of the racemate. I was sensible, and managed to do several technical chores in a reasonable way. Easy sleep at 15 hours into it.
(with 2.3 mg of the "R" isomer) The water solution of the hydrochloride salt has a slightly sweetish taste! I was at a +++ without question, but there was a slight down mood towards the end. And it lasted a really long time; I was distinctly aware of residual stuff going on, well into the next day.
(with 6.3 mg of the "S" isomer) I was at a benign one-and-a-half plus at about two hours, and finally flattened out at a ++. Would I double this dose? Probably not, but half again (to 9 or 10 milligrams) would feel safe for a plus 3. By evening I was near enough baseline to drive into town for a social obligation, but even when trying to sleep later that night there was some residue of imagery; remarkably, it was all in slow motion. The fantasies were slow-paced and sluggish. It would have been interesting to have explored eyes-closed during the day.
EXTENSIONS AND COMMENTARY: Again, as with every other psychedelic amphetamine analogue which has a chiral center and has been explored as the individual optical isomers, it is the "R" isomer that is the more potent. And again, the other isomer, the "S" isomer, still shows some activity. The same was true with DOB, and DOM, and MDA. The only exception was MDMA, but then that is more of a stimulant, and there is virtually no psychedelic component to its action. Rat studies, where there is a measure of the discrimination of a test compound from saline, have shown the "R" isomer to have about twice the potency of the "S" isomer. That the "R" is more potent is certain, but the above reports would suggest that the factor would be closer to times-four rather than times-two.
A number of studies with DOI in animal models have shown it to have an extremely high binding capacity to what are called the 5-HT2 receptors. Serotonin is a vital neurotransmitter in the brain, and is strongly implicated in the action of all of the phenethylamine psychedelics. The place where it acts, at the molecular level, is called its receptor site. As an outgrowth of the cooperative studies of the medicinal chemists working closely with the neuropharmacologists, a number of compounds have emerged that interact with these sites. But this one interacts with these sites and not those, and that one interacts with those sites and not these. So, there has developed a collection of sub-divisions and sub-subdivisions of receptor sites, all related to serotonin, but each defined by the particular compound that interacts most tightly with it.
Thus, there were serotonin "1" receptors, and then there were "1" and "2" receptors, and then "1a" and "1b" and "2a" and "2b" receptors, and on and on. These are called 5-HT receptors, since the chemical name for serotonin is 5-hydroxytryptamine, and the scientist would never want to let the layman know just what he is talking about. DOI has been synthesized with a variety of radioactive iodine isotopes in it, and these tools have been of considerable value in mapping out its brain distribution. And by extrapolation, the possible localization of other psychedelic compounds that cannot be so easily labelled. A small neurochemical research company on the East Coast picked up on these properties of DOI, and offered it as a commercial item for research experiments. But I doubt that they are completely innocent of the fact that DOI is an extremely potent psychedelic and that it is still unrecognized by the Federal drug laws since, in their most recent catalog, the price had almost doubled and a note had been added to the effect that telephone orders cannot be accepted for this compound.
The four-carbon butylamine homologue (the ARIADNE analogue) of DOI has been synthesized. A mixture of the free base of 1-(2,5-dimethoxyphenyl)-2-aminobutane (see preparation under DOB) and phthalic anhydride was fused, cooled, and recrystallized from either methanol or cyclohexane to give crystals of N-[1-(2,5-dimethoxyphenyl)-2-butyl]phthalimide with a melting point of 76-77 °C and an analysis (C20H21NO4) C,H,N. This was iodinated with iodine monochloride in acetic acid to give N-[1-(2,5-dimethoxy-4-iodophenyl)-2-butyl]phthalimide which was chromatographically distinct from the uniodinated starting material (silica gel, CH2Cl2 ), but which did not crystallize. This was treated with hydrazine hydrate in ethanol to provide 1-(2,5-dimethoxy-4-iodophenyl)-2-aminobutane hydrochloride which was crystallized from CH3CN/EtOH to give white crystals with a mp of 217-218.5 °C and an analysis (C12H19CINO2) C,H,N. This butyl homolog of DOI has been assayed at up to four milligrams, and is without any central effects whatsoever. An experiment with 12.4 microcuries of 131I labelled material with the whole body scanner showed most of it accumulating in the gut and liver, with almost none to the brain.
For those who find such statistics interesting, the parent compound DOI vies with DOB as probably the most potent of the phenethylamine psychedelics as of the moment, and certainly one of the most long lived.
A very important, centrally pivotal, and completely paradoxical compound in this area, is the N,N-dimethyl homologue of DOI, or 2,5-dimethoxy-N,N-dimethyl-4-iodoamphetamine (IDNNA). This compound was the starting point of the study of a large number of homologues and it deserves, and has received, a separate recipe.
| [ |
[Main Index] | [Forward |

