
#57 DME
3,4-DIMETHOXY-beta-HYDROXYPHENETHYLAMINE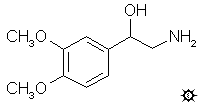
|
| [3D .mol structure] |
A well-stirred suspension of 4.7 g LAH in 500 mL anhydrous Et2O was brought up to a gentle reflux, and 4.7 g 3,4-dimethoxy-a-hydroxyphenylacetonitrile was leached in from a Soxhlet thimble, over the course of 3 h. The color of the ether solution progressed from yellow to green, to an eventual blue. The reflux was maintained for 16 h. After cooling again, there was added (carefully) a solution of 27 g H2SO4 in 500 mL H2O. The completely clear two-phase mixture was separated, and the aqueous phase treated with 87 g potassium sodium tartrate. The addition of 25% NaOH brought the pH >9, and this phase was extracted with 4x100 mL CH2Cl2. Removal of all the organic solvents under vacuum gave a residue that was part oil and part solid. This was extracted with 4x50 mL boiling Et2O, the extracts pooled, and saturated with anhydrous HCl gas. The 0.95 g of pale-yellow crystals that formed were removed by filtration, and finely ground under 5 mL CH3CN. There remained, after refiltration and air drying, 0.85 g of 3,4-dimethoxy-beta-hydroxyphenethylamine hydrochloride, DME, with a mp of 170-172 °C.
DOSAGE: greater than 115 mg.
DURATION: unknown.
QUALITATIVE COMMENTS: (with 115 mg) I was faintly nauseous about an hour after taking the compound, and perhaps I was more alert than usual in the evening. Substantially no effects.
EXTENSIONS AND COMMENTARY: The rationale for exploring the beta-hydroxylated phenethylamines, especially those with oxygens at the biologically important 3- and 4-positions, has already been presented. Norepinephrine is a beta-hydroxylated phenethylamine with oxygens at these two ring positions. With DME, these are masked as two methyl ethers, and the initials DME stand for 3,4-dimethoxyphenyl-beta-ethanolamine. This is an alternate name for 3,4-dimethoxy-beta-hydroxyphenethylamine.
An exactly analogous compound is 3,4-methylenedioxy-beta-ethanolamine, where the masking is done with the biologically more fragile methylenedioxy ether. Originally I had called this compound MDE (methylenedioxyethanolamine) but that code has been, since 1975, used exclusively for 3,4-methylenedioxy-N-ethylamphetamine, which is a recipe all by itself. Under the discussion of members of the BOX series, there is a methylenedioxyphenethylamine with a methoxyl group at the beta-position, and it is called BOH (q.v.). There, a reasonable code name for this specific compound is given, namely BOHH. RBOS stands for the beta-oxygen function on a phenethylamine; this is the heart of the BOX family. The RHS which is the third letter of BOHH stands for the free hydroxyl group. And the final RHS is for homopiperonylamine (which is the trivial name for the compound without the hydroxyl group). BOHH, or 3,4-methylenedioxy-beta-hydroxyphenethylamine, or 3,4-methylenedioxy-beta-ethanolamine, has also be assayed in man at up to 100 milligrams without any effects, and must be considered, as of now, to be inactive centrally. The possible toxic roles of beta-ethanolamines as potential adrenolytic agents, have been discussed in the BOHD recipe. And beware of the use of the code name MDE in the very old literature. It might be this BOHH compound.
| [ |
[Main Index] | [Forward |

