
#23 2C-D
LE-25; 2,5-DIMETHOXY-4-METHYLPHENETHYLAMINE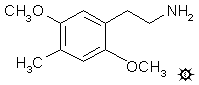
|
| [3D .mol structure] |
A mixture of 34.5 g POCl3 and 31.1 g N-methylformanilide was heated for 10 min on the steam bath, and then there was added 30.4 g of 2,5-dimethoxytoluene. Heating was continued for 2.5 h, and the viscous, black, ugly mess was poured into 600 mL of warm H2O and stirred overnight. The resulting rubbery miniature-rabbit-droppings product was removed by filtration and sucked as free of H2O as possible. The 37.2 g of wet product was extracted on the steam-bath with 4x100 mL portions of boiling hexane which, after decantation and cooling, yielded a total of 15.3 g of yellow crystalline product. This, upon recrystallization from 150 mL boiling hexane, gave pale yellow crystals which, when air dried to constant weight, represented 8.7 g of 2,5-dimethoxy-4-methylbenzaldehyde, and had a mp of 83-84 °C. Anal. (C8H12O3) C,H,N. The Gattermann aldehyde synthesis gave a better yield (60% of theory) but required the use of hydrogen cyanide gas. The malononitrile derivative, from 5.7 g of the aldehyde and 2.3 g malononitrile in absolute EtOH, treated with a drop of triethylamine, was an orange crystalline product. A sample recrystallized from EtOH gave a mp of 138.5-139 °C.
A solution of 8.65 g 2,5-dimethoxy-4-methylbenzaldehyde in 30 g nitromethane was treated with 1.1 g anhydrous ammonium acetate and heated for 50 min on the steam bath. Stripping off the excess nitromethane under vacuum yielded orange crystals which weighed 12.2 g. These were recrystallized from 100 mL IPA providing yellow crystals of 2,5-dimethoxy-4-methyl-beta-nitrostyrene which weighed, when dry, 7.70 g. The mp was 117-118 °C, and this was increased to 118-119 °C upon recrystallization from benzene/heptane 1:2.
To a well stirred suspension of 7.0 g LAH in 300 mL of warm THF under an inert atmosphere, there was added 7.7 g 2,5-dimethoxy-4-methyl-beta-nitrostyrene in 35 mL THF over the course of 0.5 h. This reaction mixture was held at reflux for 24 h, cooled to room temperature, and the excess hydride destroyed with 25 mL IPA. There was then added 7 mL 15% NaOH, followed by 21 mL H2O. The granular gray mass was filtered, and the filter cake washed with 2x50 mL THF. The combined filtrate and washes were stripped of their volatiles under vacuum to give a residue weighing 7.7 g which was distilled at 90-115 °C at 0.3 mm/Hg to provide 4.90 g of a clear, white oil, which crystallized in the receiver. This was dissolved in 25 mL IPA, and neutralized with concentrated HCl which produced immediate crystals of the salt. These were dispersed with 80 mL anhydrous Et2O, filtered, and washed with Et2O to give, after air drying to constant weight, 4.9 g of fluffy white crystals of 2,5-dimethoxy-4-methylphenethylamine hydrochloride (2C-D). The mp was 213-214 °C which was not improved by recrystallization from CH3CN/IPA mixture, or from EtOH. The hydrobromide salt had a mp of 183-184 °C. The acetamide, from the free base in pyridine treated with acetic anhydride, was a white crystalline solid which, when recrystallized from aqueous MeOH, had a mp of 116-117 °C.
DOSAGE: 20 - 60 mg.
DURATION: 4 - 6 h.
QUALITATIVE COMMENTS: (with 10 mg) There is something going on, but it is subtle. I find that I can just slightly redirect my attention so that it applies more exactly to what I am doing. I feel that I can learn faster. This is a `smart' pill!
(with 20 mg) Butterflies in stomach whole time. OK. This is about the right level. In retrospect, not too interesting. Primarily a stimulant, not entirely physically pleasant. The visual is not too exciting. I am easily distracted. One line of thought to another. I feel that more would be too stimulating.
(with 30 mg) I was into it quite quickly (not much over three-quarters of an hour) and got up to a ++ by the end of an hour. There is something unsatisfactory about trying to classify this level. I had said that I was willing to increase the dose to a higher level, to break out of this not-quite-defined level into something psychedelic. But I may not want to go higher. Under different circumstances I would not mind trying it at a considerably lower dosage, perhaps at the 10 or 15 milligrams. I do not have a comfortable label on this material, yet.
(with 45 mg) There was a rocket from the half-hour to the one and a half hour, from nothing up to a +++. Somehow the intimacy and the erotic never quite knit, and I feel that I am always waiting for the experience to come home. Talking is extremely easy, but something is missing. Appetite is good. I am down by the fifth hour, and sleep is comfortable. This compound will take some learning.
(with 75 mg) This is a +++, but the emphasis is on talking, not on personal interacting. I am putting out, but my boundaries are intact. I was able to sleep at the sixth hour. Communication was excellent. This is fast on, but not too long lived. Maybe a therapy tool?
(with 150 mg) A truly remarkable psychedelic, one which could compare favorably with 2C-B. There are intense colors, and I feel that more would be too much.
EXTENSIONS AND COMMENTARY: Wow! This particular compound is what I call a pharmacological tofu. It doesn't seem to do too much by itself, always teasing, until you get to heroic levels. But a goodly number of experimental therapists have said that it is excellent in extending the action of some other materials. It seems to boost the waning action of another drug, without adding its own color to the experience. Yet, the comment above, on the high level of 150 milligrams, is a direct quote from the use of this compound in Germany (where it is called LE-25) in therapeutic research.
This is probably the most dramatic example of the loss of potency from an amphetamine (DOM, active at maybe 3 milligrams) to a phenethylamine (only one tenth as active). It is so often the case that the first of a series is not the most interesting nor the most potent member. As intriguing and as difficult-to-define as the 2C-D story might be, the next higher homologue of this set, 2C-E, is maximally active at the 15 to 20 milligram level, and is, without any question, a complete psychedelic.
The N-monomethyl and the N,N-dimethyl homologues of 2C-D have been synthesized from 2C-D. The N-monomethyl compound was obtained by the quaternization of the Schiff's base formed between 2C-D and benzaldehyde with methyl sulfate, followed by hydrolysis; the hydrochloride salt had a melting point of 150-151 °C, from EtOH. The N,N-dimethyl compound resulted from the action of formaldehyde-formic acid on 2C-D; the hydrochloride salt had a melting point of 168-169 °C from EtOH/ether. These two compounds were some ten times less effective in interfering with conditioned responses in experimental rats. There is no report of their having been explored in man.
[ Erowid Addition: Darrell Lemaire's Contribution # : In January, 2015, chemist and psychonaut Darrell Lemaire announced that he had decided to come out publicly as the author "Lazar" who conducted the "extensive study" referenced in the following paragraph. For many years, Mr. Lemaire had preferred to remain anonymous, but the Shulgin Team is helping add credit for his work back into the literature. See Darrell Lemaire Character Vault.]
I have learned of an extensive study of ethoxy homologues of a number of the phenethylamines in the 2C-X series; they have been collectively called the "Tweetios." This Sylvester and Tweety-bird allusion came directly from the compulsive habit of trying to alleviate the boredom of driving long distances (not under the influence of anything) by the attempt to pronounce the license plates of cars as they passed. The first of this series of compounds had a name that indicated that there was an ethoxy group at the 2-position, or 2-EtO, or Tweetio, and the rest is history. In every compound to be found in the 2C-X family, there are two methoxy groups, one at the 2-position and one at the 5-position. There are thus three possible tweetio compounds, a 2-EtO-, a 5-EtO- and a 2,5-di-EtO-. Those that have been evaluated in man are included after each of the 2C-X's that has served as the prototype. In general, the 2-EtO- compounds have a shorter duration and a lower potency, the 5-EtO- compounds have a relatively unchanged potency and a longer time duration; the 2,5-di-EtO- homologues are very weak, if active at all.
The 2-EtO-homologue of 2C-D is 2-ethoxy-5-methoxy-4-methylphenethylamine, or 2CD-2ETO. The benzaldehyde (2-ethoxy-5-methoxy-4-tolualdehyde) had a melting point of 60.5-61 °C, the nitrostyrene intermediate a melting point of 110.5-111.5 °C, and the final hydrochloride a melting point of 207-208 °C. The hydrobromide salt had a melting point of 171-173 °C. At levels of 60 milli-grams, there was the feeling of closeness between couples, without an appreciable state of intoxication. The duration was about 4 hours.
The 5-EtO- homologue of 2C-D is 5-ethoxy-2-methoxy-4-methylphenethylamine, or 2CD-5ETO. The benzaldehyde (5-ethoxy-2-ethoxy-4-tolualdehyde) had a melting point of 81-82 °C, and the details of this synthesis are given in the recipe for IRIS. The nitrostyrene intermediate had a melting point of 112.5-113.5 °C and the final hydrochloride salt had a melting point of 197-198 °C. The hydro-bromide salt had a melting point of 158-159 °C. At dosage levels of 40 to 50 milli-grams, there was a slow, gradual climb to the full effects that were noted in about 2 hours. The experience was largely free from excitement, but with a friendly openness and outgoingness that allowed easy talk, interaction, humor, and a healthy appetite. The duration of effects was 12 hours.
The 2,5-di-EtO- homologue of 2C-D is 2,5-diethoxy-4-methylphenethylamine, or 2CD-2,5-DIETO. The benzaldehyde (2,5-diethoxy-4-tolualdehyde) had a melting point of 102-103 °C, the nitrostyrene intermediate a melting point of 108-109 °C, and the final hydrochloride salt a melting point of 251-252 °C. At a level of 55 milligrams, a plus one was reached, and what effects there were, were gone after four hours.
| [ |
[Main Index] | [Forward |

