Step 1. (4 hours)
Distillation of your essential oil to obtain pure safrole.
Written by Chromic (updated May 13, 2002), with thanx going out to Bright Star for the document which this is liberally based on.
Materials required for this step:
- Essential oil that contains safrole (Chinese or Brazilian Sassafras, Brown Camphor etc.)
- Complete ground-glass jointed vacuum fractional distillation setup.*
- Heating and stirring equipment**
- Vacuum source***
- Cooling water source****
- Column insulation
* You should have 24/40 or 19/22 500mL & 250ml RB flasks, 200mm Vigreux column, 3-way adapter, Liebig condenser, with thermometer (and thermometer adapter, if necessary), stands, clamps and bossheads.
** Two options exist for a heating/stirring equipment. Either you can use a stirring hot plate, stir bar, aluminum pan, cooking oil and thermometer or an electric heating mantle, stirbar, variac and (hotplate) stirrer.
*** Three options exist for a vacuum source. Either you can use a professional vacuum pump, a converted refrigerator compresser or a water aspirator.
**** Two options exist for a water source. Either you can run fresh tap water through vinyl tubing to your condenser and back to the drain, or you can use a small recirculating water pump such as those found in water fountains.
Procedure:
Time to set up for a fractional vacuum distillation (see notes unless you've already performed this technique!).
There are two primary methods of heating the oil in the flask. One simple method for heating the oil has the clamped flask sitting on an electric heating mantle that is controlled by a variac. The heating mantle rests on a stirrer (or a hot plate stirrer although the heat is never turned on). An alternative method is to have the clamped flask sitting in an aluminum saucepan. This saucepan rests on the hot plate stirrer. Fill the saucepan with enough oil to mostly submerge your 500mL distilling flask, and use a cooking oil thermometer to monitor the temperature of the oil bath.
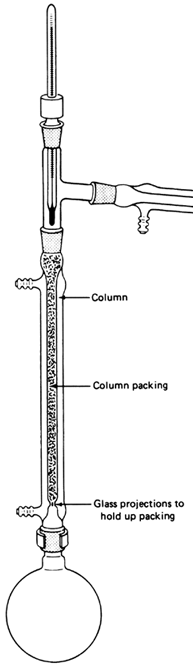
You should have no more than 300ml of our fragrant essential oil in the 500mL round bottom flask and the flask should also contain a 1" egg (or octagonal) stirbar. Add your column to the flask, then place the 3-way adapter ontop, then the condenser, then the vacuum adapter, and put a 250mL RB flask as the receiving flask. You should have placed a clamp at the fractionating column and at the receiving flask. If desired, you can optionally place another clamp on the condenser. Last fit your thermometer to your thermometer adapter (if you're using one) and place your thermometer into the 3-way adapter. Get your vacuum ready, but not connected or turned on.
During this run, you're going to have to insulate your fractionating column. Glass wool is the best heat resistant insulator. If you find it at your hardware store make use of it. Fleece also works incredibly well, but will not tolerate temperatures above 150°C. If you expect your oil to come across above that temperature and can't find glass wool, cotton cloth (such pieces of a towel or sock), wrapped around the column will suffice. Use wire to keep the insulating material on. If you are distilling your safrole at a higher temperature you will need insulation, otherwise too much safrole will fall back into the distillation pot as reflux. If you have a good vacuum (<10mmHg) or your column is silvered and vacuum jacketed you likely can get away with no insulation.
Now set your stirrer to spin like mad and heat the oil bath up quickly to the temperature you expect your safrole to come over. Use the oil thermometer to monitor the temperature of your oil bath, so you don't vastly overshoot the mark where safrole is going to come over. In my most recent case it was 145°C for a 60mmHg vacuum. Sometimes I'll just set it on low, and go off to grab something to eat as the oil warms up. Now, when the cooking oil is at 145°C... lower the heat, and apply the vacuum. Nothing usually comes over. Now, continue heating, but this time slowly, to about 25-30°C above that temperature... nothing usually comes over. If you have got something to come over, and it isn't your main fraction, remove the heat, disconnect your vacuum, and empty the garbage fraction that's in your flask, then replace it and continue... All of a sudden you're going to see the temperature on your thermometer climb quickly and hit that temperature, say 145°C, and you will hold it there... do not rush this step of the distillation... and now just slowly collect the oil (1-2 drops/second) until no more oil starts to come over, and you've shot at least 5°C past that temperature. So in my case I collect 145-150°C. With more experience, you can narrow that range to get even more pure safrole. A good visual indicator of a different fraction starting to come over, is there will be little "swirlies" in the receiving flask as the new liquid drips in. This is because the index of refraction of the next fraction is different.
Make a note of the temperature that your safrole comes over at.
If the temp that your safrole comes over is higher than 160°C check your seals on the tubing and glassware... more than likely there is a little leak. If you're using an aspirator, and if you don't have good water pressure, you won't be able to get a good vacuum. Also, remember the Vaseline! Use an automotive vacuum gauge setup on a T-connector of your vacuum tubing if you suspect a leak, this will be able to quickly let you know if you're reaching near full vacuum each time. You should be getting at a very minimum, 28" of vacuum, and preferably higher.
At the end of the distillation, you should have a water white oil that really refracts light... and has a lovely smell... a little like licorice, a little like rootbeer, a nice potpourri smell... expect a yield of 75-90% safrole.
Now you have the fun job of cleaning the dishes. The best way is to use acetone to remove all the oils then wash with soapy water (if you feel it necessary). If there is any charring in your distilling flask, rinse with acetone, then let it soak in household ammonia or 10% sodium hydroxide for a few hours, then use a brush to easily scrub it out.
Notes
Steps on building before you jump in:
Figure 1:
Example of a simple distillation setup.

Step 1: Once you get your distillation apparatus, set up your distillation apparatus on your bed before you try to put it together on the stand. You will get a good idea about how the pieces go together, and become familiar with the fragility of the rig.
Step 2: Now you're ready to move it to the stand. Remember, this thing is build like a house.. bottom up. The boss-heads connect to the 3/8" or 1/2" steel rods that stand is made of and keep the clamps in place, the clamps will attached on both of the round bottom flasks, the column and perhaps on the condenser as well. Read Zubrick for more advice if you need more assistance.
Step 3: Get practice distilling regular tap water using an oil bath. Make sure your condenser water is reliable whether is comes from a recirculating fountain pump or from the tap, do not try to siphon water thru the condenser instead of using a pump or tap, although this technique works, it's too inconvenient and requires too much attention. Once you've done this, setup for regular fractional distillation and proceed.
The oil bath idea is simple... a metal bowl (do not use steel if you're using magnetic stirring as your stirrer will not function properly) with a flat bottom rests on the hotplate. It is filled with peanut or safflower oil. The distillation flask sits in the bowl but not touching the bottom, so that the hotplate heats the bowl, the bowl heats the oil, the oil heats the distillation flask. This is very effective, and although it will give off a bit of an oil smell, it's tolerable. And will be perfect for all your distillation needs... most especially if you do it under vacuum. You want even heating, especially if you're not using magnetic stirring. Be warned, if you're not using a vacuum source to do this distillation I recommend not using this method. The oil will smoke. Either use a heating mantle or a flat-bottomed flask that is "tented" with aluminum foil. Rest the FB flask directly on the hot plate. Be very careful to remove the heat once the distillate it collected if you use this method, or you will scortch your flasks. While on the subject of flat bottomed flasks, it should be mentioned to NEVER use a large (>250ml) flat bottom flask under vacuum. The risk of implosion is quite high.
The water should come across at 100°C, otherwise this indicates that your thermometer is off or the atmospheric pressure is significantly off in your area.
Step 4:Try again with water, but now under vacuum, and note the temperature it comes over at.
Always put a little Vaseline on the ground glass joints when doing a vacuum or high temperature distillation... this way they won't stick when you try to take them apart and will also let you know if you have a leak (air bubbles will be seen travelling thru the vaseline if you do have one). If you forget this advice and your joints freeze together, free the stuck joints by evenly heating the outer joint with a lighter or propane torch. The glass should expand and you should be able to free each piece of glass.
This distillation with water will indicate the strength of your vacuum (see vacuum distillation notes at the end of this document) Once you know what your vacuum source can pull, then you can cross-reference it to when safrole should come over to know exactly what to keep and what to throw out when you do the distillation.
Remember, the flask is never directly heated in vacuum distillation, it can be heated with an "air bath" for regular distillation but it is far too erratic for vacuum distillation.
Step 5: You're now ready for sassafras! Go up and continue reading from the top. [Erowid Note: Edited to clarify that the oil here is NOT safrole based on reader feedback. July 2007.] If you DON'T think you're ready for sassafras, go buy some cheaper oil of star anise, and practice with it. Star Anise's oil, anethole, has very similar physical properties that makes it useful for practicing techniques. It will come across at almost the same temperature as safrole. If you want to experiment more with the technique of fractional distillation, try distilling vodka or wine. Please note that MDMA synthetic techniques using anethole can result in dangerous products that are not MDMA. Anethole is NOT a substitute for safrole.
Other notes
If you do not have a vigreux column:
Although it is preferable to use a short Vigreux column, distillation without a column will suffice as so long as you have magnetic stirring.
If you do not have the magnetic stirring:
If you do not have magnetic stirring, it will be much harder to do this distillation, but it is still possible. You must use a column or a Claisen adapter so that if you experience any heavy bumping, that the liquid will not spill over into the receiving flask. If you do not use magnetic stirring, add about 5g of very porous boiling stones made by breaking up pumice stone.
You can use room temperature water for your condenser. Note that four clamps are used in this setup, one at the distilling flask, one to secure the fractionating column, one to secure the condenser, one to secure the receiving flask. It is a good idea to use at least 3 clamps to hold this setup, any less is unsturdy. In this setup there is no thermometer adapter, as the three way adapter has one built in. Note the position of the thermometer bulb, this is where you want to place your thermometer. In this example boiling stones are shown being used, however it is more likely that you will be using magnetic stirring.
Figure 2:
Example of a fractional distillation setup.
Vacuum distillations notes:
Boiling point data of Safrole, isosafrole and water (adapted from Niels Bohr, as posted at The Hive). Note that isosafrole is not an intermediate in the Wacker oxidations to obtain ketone, only in the peracid (performic, peracetic, Oxone) methods to obtain ketone.
mmHg is a term referring to the absolute pressure in torr, where:
760 mmHg = 760 torr = 1 atm = 0 psig
0 mmHg = 0 torr = 0 atm = -14.7 psig
Inches of mercury refers to a gauge measurement usually seen on automotive gauges. It is relative to the atmospheric pressure, where:
0" Hg = 1 atm = 760 torr = 760 mmHg = 0 psig
29.92"Hg = 0 atm = 0 torr = 0 mmHg = -14.7psig.
Pressure (mmHg/Torr) |
Vacuum (Inches Hg) |
Safrole bp °C |
Isosafrole bp °C |
Water bp °C |
|
760 700 600 500 400 300 200 100 90 80 70 60 50 40 30 20 15 10 8 6 5 0 |
0" 2.36" 6.30" 10.24" 14.17" 18.11" 22.05" 25.98" 26.38" 26.77" 27.17" 27.56" 27.95" 28.35" 28.74" 29.13" 29.33" 29.53" 29.61" 29.69" 29.72" 29.92" |
234.5 231.0 224.7 217.3 208.4 197.7 183.7 162.2 159.2 155.7 152.1 148.0 143.3 137.3 130.0 120.8 114.4 106.4 102.0 96.6 93.2 ~ |
255.0 251.2 244.6 236.7 227.6 216.3 201.8 179.1 175.9 172.2 168.4 164.2 159.2 153.0 145.2 135.6 129.0 120.5 116.0 110.2 106.6 ~ |
100.0 97.7 93.5 88.7 83.0 75.9 66.4 51.6 49.4 47.1 44.5 41.5 38.1 34.1 29.0 22.1 17.5 11.2 7.9 3.8 1.2 ~ |