Synthesis of a Clonitazene/Etonitazene Analog[ Back to the Chemistry Archive ] IntroductionThe compound described below ("Nitazene") is a simpler designer analog of the very strong opioids Clonitazene and Etonitazene, the latter being about 1000 times more potent than morphine(!). The synthesis can easily be adapted for the synthesis of the above compounds by exchanging the benzyl cyanide in the first step to p-chlorobenzyl cyanide (for Clonitazene) or p-ethoxybenzyl cyanide (for Etonitazene). 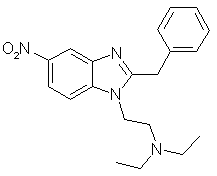
2-benzyl-5-nitro-benzimidazoleA solution of 17.5 grams of benzyl cyanide and 8.7 grams of ethanol is saturated with dry hydrogen chloride while stirring and cooling with ice water; the mixture is allowed to stand for 12 hours at room temperature and then stirred with a suspension of 15.3 grams of 2-amino-4-nitroaniline in 200 ml of dioxane. The reaction mixture is stirred for 24 hours at about 85°C, then boiled under reflux for four hours and finally, when still hot, rendered sharply acid with a mixture of ethyl acetate and hydrochloric acid. The hydrochloride of 2-benzyl-5-nitro-benzimidazole which crystallizes out is purified by recrystallization from dilute aqueous hydrochloric acid with the aid of animal charcoal. By adding aqueous ammonia, the free base is obtained which after recrystallization from a mixture of dioxane and ligroin melts at 189°C. 1-(N,N-diethylamino-ethyl)-2-benzyl-5-nitro-benzimidazole ("Nitazene")A solution of 68 grams of sodium ethylate in ethanol, as concentrated as possible, is added dropwise to 253 grams of 2-benzyl-5-nitro-benzimidazole in 3.0 litres of dioxane at 60°C with stirring. After the addition is complete, the sodium salt of 2-benzyl-5-nitrobenzimidazole, which has crystallized in the form of fine yellow needles, is suction-filtered and dried in vacuo at 60°C. To 27.6 grams of this sodium salt suspended in 600 ml of absolute dioxane there are added dropwise 13.5 grams of N,N-diethylamino-ethyl chloride at 60°C. Stirring is carried on for an hour at 60°C, the sodium chloride formed is filtered off, and the filtrate is evaporated. The resulting mixture of 1-(N,N-diethylamino-ethyl)-2-benzyl-6-nitro-benzimidazole and 1-(N,N-diethylamino-ethyl)- 2-benzyl-5-nitrobenzimidazole is separated into its two isomers in the following manner: After crystallizing the product twice from ether there is obtained pure 1-(N,N-diethylamino-ethyl)-2-benzyl-6-nitro-benzimidazole melting at 96-97°C. The mother liquor is evaporated and in ethanol the calculated quantity of ethanolic hydrochloric acid is added. The precipitated hydrochloride, after being repeatedly recrystallized from water, gives pure 1-(N,N-diethylamino-ethyl)-2-benzyl-5-nitro-benzimidazole hydrochloride melting at 199-201°C. Instead of the sodium ethylate solution there can be used a solution of sodamide in liquid ammonia (prepared from 200 ml of ammonia, 2.3 grams of sodium and 0.06 gram of ferric nitrate) for the condensation. Reference: Example 6 from US patent 2,935,514. |