MAB and DABD - Friends of GHBby Methaco(s)mic[ Back to the Chemistry Archive ] I have synthesized and bioassayed two relatives of GHB, MAB (Methyl 4-Acetoxy-Butanoate) and DABD (1,4-DiAcetylButaneDiol). I don't claim to be the first or anything, but since I can't find any of those compounds described here before I feel I have to bring the subject up. The MAB synthesis can be found in several patents and DABD is such an obvious compound that I'm sure it has been made and tested before. I performed the synthesizes and the assays well over a year ago. 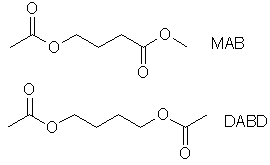
MAB Synthesis: 500ml 99.8% MeOH and 4ml 98% sulfuric acid was mixed in a 1000ml Erlenmeyer flask, 75ml 99% gamma-butyrolactone was added. The flask was plugged and left in room temperature for one hundred days (!) (five days in the patent). Sodium carbonate was added carefully until the foaming stopped, 10g were needed. 20g anhydrous sodium sulfate was added to dry the solution. The solution was concentrated under vacuum in a 1000ml RB. When all MeOH was gone 300ml dH2O was added. The solution was extracted with 3x150ml chloroform. The chloroform was dried with 13g of anhydrous magnesiumsulfate. The chloroform was removed by distillation and reused. The concentrate was placed in a 1000ml Erlenmeyer flask and cooled with ice/salt, when cold (~0°) 80ml 98% acetic anhydride and 100ml 99% pyridine was added. The flask was plugged and I stirred at 1000rpm with magstirrer. After 14 hours a mixture of 100ml dH2O, 200g ice and 100ml concentrated ACS hydrochloric acid that had been prepared in a 500ml beaker was slowly added to the Erlenmeyer flask. The mixture was stirred until it reached room temperature and was extracted with 4*100ml chloroform (two layers formed even before the chloroform was added). The organic layer was treated with 2*100ml saturated sodium bicarbonate solution and then with 2*100ml saturated sodium chloride solution. 11g anhydrous magnesiumsulfate was used to dry the chloroform extract. The chloroform was removed by distillation and the MAB was distilled at aspirator vacuum several times to get it very pure. I must have been really stupid >1 year ago because I can't find the boiling point in my notes, but it was perhaps 120°C something (in the patent the boilingpoint is 56°C at 0.1 torr). The story above is very much what you can read in the patents, very straightforward and no surprises. The yield was 30-35ml pure product. I lost some in the repeated distillations. This is not the kind of drug you make to get rich anyway. References:
MAB Bioassay: 0.5g (~0.5ml) is the lowest dose that can be felt, 1.1-1.6g (~1.1-1.6ml) is a useful dose range and 1.8g (~1.8ml) and above is too much. If taken on empty stomach the effects can be felt within 5 to 10 minutes. Intoxication similar to GHB develops. The intoxication is not by far as nice as the one you get with GHB, in fact it can be quite unpleasant on higher doses (>1.8-1.9g). While GHB just puts you to sleep this material is much more annoying on higher doses and the initial intoxication will last up to 5-6 hours on a (too) high dose. In the 1.1.-1.6g range the effects are much nicer, mild intoxication that lasts only for 2-3 hours but you will be deeply relaxed and experience anxiolytic effect as long as 10-12 hours after consumtion. Lower doses (<<1g) feels like 5mg diazepam, not much that is, but makes you calm and relaxed for several hours. Since this material is not particularly fun or nice when overdosed (not for me anyway) it could be a good daytime anxiolytic with a rather low abuse potential. DABD Synthesis: 20ml 99% 1,4-butanediol was introduced in a 100ml RB, 45ml 98% acetic anhydride was added, it didn't even heat up so I brought it to reflux and held it there for 30 minutes. Dumped the mixture in 200ml dH2O and extracted it with 2*70ml chloroform. Treated the chloroform extracts with 2*50ml saturated sodium carbonate solution. Dried the organic layer over anhydrous magnesiumsulfate and removed the chloroform by distillation. The DABD was distilled with aspirator vacuum (stupid me! no boiling point, if I redistill what's left I can inform the interested) the yield was ~25ml. DABD smells rather nice; it smells like crushed leaves from roses! DABD Bioassay: I tried this more recently so the following is much more accurate then the account above. I have only used doses between 1.0 and 1.5ml. 1.0ml is not felt much and 1.5ml is like a medium dose of GHB. Ordinary 1,4-butanediol "BD" feels almost exactly as GHB, can we all agree on that? So what does acetylation do to the effects? It can be felt after 5-10 minutes (tastes like gasoline) the peak of effects occur at the 40 minutes point and then the intoxication gradually dissipates during the following hours and at the 3 hour point the intoxication is over. Perhaps a weak sedation and muscle relaxation lasts to the 5-hour point. The effects are GHB like but not as nice and not euphoric, nothing sexual and the disinhibition is minimal. Relaxing and anxiolytic properties, but not fun. Rather much like MAB perhaps, but without the nice long-lasting positive effects. My stomach doesn't like this stuff I feel like I have to burp but I can't. -Methaco(s)mic |