Summary
[3H]Methcathinone (2-[3H]methylamino-1-phenylpropan-1-one), an inhibitor of monoamine reuptake transporters, was synthesized by N-methylation of cathinone using [3H]methyl iodide in toluene/methanol. A two-step purification procedure employing preparative silica gel TLC, followed by reverse-phase HPLC, was developed to separate [3H]methcathinone from unreacted starting material and side products. We were able to generate radio- and chemically pure [3H]methcathinone, ready for use in pharmacological experiments, in about eight hours. Overall yield was about 4% based on [3H]methyl iodide.
Introduction
Methcathinone (2-methylamino-1-phenylpropan-1-one; Fig. 1) is the N-methyl derivative of cathinone, a naturally occurring psychostimulant found in the leaves of Catha edulis, the khat bush. Methcathinone was first synthesized in 19281, but its psychopharmacology remained unexplored until Parke, Davis, and Company developed methcathinone as an analeptic in the late 1950's2. More recently, methcathinone has been used outside of medical settings for its euphoric properties and is now designated a Schedule I Controlled Substance in the United States3,4. Behavioral studies have shown that methcathinone will substitute for cocaine or amphetamine in animals trained to discriminate either cocaine or amphetamine from saline in stimulus generalization tests5. Baboons will self-administer methcathinone, confirming that the drug has reinforcing properties6. Together, these data suggest that methcathinone may be acting on monoaminergic systems, and we have recently completed neurochemical studies demonstrating that methcathinone is indeed a potent inhibitor of catecholamine transport with more modest effects on serotonin uptake7. To further our understanding of the mechanism of action of methcathinone, we synthesized [3H]methcathinone as a potential probe for the amphetamine binding site on the reuptake carriers. We also developed an efficient separation and purification protocol employing preparative thin-layer chromatography and reverse-phase high-pressure liquid chromatography that allowed us to synthesize and isolate radio- and chemically pure [3H]methcathinone in less than one day.
Materials and Methods
Reagents
Cathinone was synthesized via the potassium permanganate oxidation of phenylpropanolamine using the procedure of Zhingel et al.8. Unlabeled methcathinone for methods development was produced using the same procedure except ephedrine was used as the starting material. All analytical data for cathinone and methcathinone (NMR, IR, melting point, and derivative formation) were consistent with the assigned structures.
Preparative TLC
Using unlabeled compounds, a TLC system was developed that allowed the separation of methcathinone from cathinone. The mobile phase consisted of n-butanol:acetic acid:water, 4:1:1. The stationary phase was a 0.25 mm glass-backed silica gel plate (Kieselgel 60 F254, 100 mm x 200 mm; Merck, Darmstadt, GBR). The test compounds were visualized as UV-quenching spots under illumination at 254 nm. Under these conditions, complete separation of methcathinone and cathinone was achieved; methcathinone migrated with Rf 0.27 and cathinone migrated with Rf = 0.44. The compounds were also visualized with 2% ninhydrin, which produces a purple color with cathinone and a reddish-orange color with methcathinone.
HPLC System
The HPLC system consisted of an Altex 210A sample injection valve with a 500 �L injection loop (Beckman, Berkeley, CA), a W-Porex reverse-phase octadecylsilane column (250 mm x 4.6 mm, 5 �m particle size; Phenomenex, Rancho Palos Verdes, CA) and 50 mM NaH2PO4 mobile phase, pH = 4.2. A Beckman Altex 110B solvent delivery module supplied eluent at a flow rate of 0.5 mL/min.; void volume was 3 mL. Compounds were detected with a Beckman 160 absorbance detector at 254 nm with output to a chart recorder (Model BD40; Kipp & Zonen, Holland). With this HPLC system, methcathinone and cathinone were baseline-resolved; cathinone eluted with tR = 18 min and methcathinone eluted with tR = 23 min. The identity of the methcathinone peak obtained from the methylation of cathinone was confirmed by spiking the HPLC sample with authentic methcathinone obtained via oxidation of ephedrine.
Experimental
Radiosynthesis of [3H]methcathinone (Fig. 1)
Cathinone hydrochloride (2.25 mg, 12.1 �mol) was added to a 200 �L Reacti-Vial (Pierce, Rockford, IL), and 20 �L methanol was added to dissolve the drug. A toluene solution (100 �L) containing 100 mCi [3H]methyl iodide (1.25 �mol, 80 Ci/mmol) was added to the vial. This was followed by the addition of triethylamine (1.74 �L, 12.5 �mol) to generate free-base cathinone in situ. The reaction vial was sealed, vortexed, and heated at 100�C for 15 min. During this time, the initially colorless solution became orange-colored. The vial was chilled in ice for a few minutes, then the entire reaction mixture was streaked onto a preparative TLC plate and the plate was dried under an airstream. The TLC plate was developed using the n-butanol/acetic acid/water mobile phase described above, then the plate was dried under a stream of warm air. The UV-quenching band corresponding to methcathinone was scraped off the plate and placed into a 1.5 mL polyethylene tube. The silica gel scrapings were extracted by suspending the silica powder in 200 �L water, vortexing, and then pelleting the silica particles at 14,000 x g for 2 min. This extraction process was repeated five times. The water extracts were combined and concentrated to about 400 �L under high vacuum at room temperature (Speed Vac Concentrator Model SVC 100H; Savant, Farmingdale, NY). The water extract was injected onto the HPLC system described above and fifty 1-min fractions were collected into glass tubes. A 1 �L aliquot of each fraction was added to 5 mL glass vials containing 3 mL scintillation fluid (Polyfluor; Packard, Meriden, CT); the vials were sealed, vortexed, and analyzed for tritium content by liquid scintillation spectroscopy using a Packard TriCarb 1600 CA. The fractions corresponding to [3H]methcathinone were stored individually at -20�C in HPLC mobile phase.
Results and Discussion
Our preliminary neurochemical studies showed that methcathinone is an inhibitor of biogenic amine accumulation7. This suggested to us that methcathinone might be a substrate for the monoamine reuptake carriers found in neuronal cell membranes We decided to synthesize radiolabeled methcathinone as a tool to determine whether it is actively transported into cells expressing the serotonin, dopamine, and norepinephrine reuptake transporters.
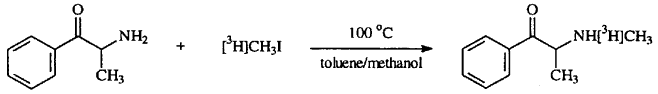
Fig. 1.
Synthetic scheme for [3H]methcathinone.
We considered a radiosynthetic route to [3H]methcathinone involving SN2 displacement of the bromine atom of 2-bromo-propiophenone by [3H]methylamine, but we abandoned this approach due to the relatively low specific activity of commercially available [3H]methylamine and the potential complication of Schiff base formation between methylamine and the benzylic ketone of 2-bromo-propiophenone. The alternate route described in this report (Fig. 1) was attractive because of the availability of the higher specific activity reagent [3H]methyl iodide. Although N-methylation of a primary amine such as cathinone with an alkyl halide can lead to undesirable side products, such as the di- and trialkylated amine, these reactions can be somewhat suppressed by using a large excess of the primary amine. Our initial experiments with unlabeled methyl iodide and tenfold molar excess of cathinone proved successful, but there were several UV-quenching side products formed despite the surplus of primary amine. In the radiosynthesis, there were several additional radioactive products formed, which were separated on the TLC plate. The UV-quenching side products migrated with the following Rf values: 0.13, 0.31, 0.55, and 0.69. Radioactive side products were observed with the following Rf values: 0.03, 0.11, 0.13, 0.15, 0.17, 0.44, 0.54. These side products were not further characterized.

Fig. 2.
Radioactivity and UV absorbance analysis of
reaction product following separation by TLC.
The TLC and HPLC systems together provided for complete isolation of the desired [3H]methcathinone from the unwanted side products. The initial separation of the reaction mixture by TLC removed most of the contaminating side-products as evidenced by the UV absorbance profile of the silica gel extract upon HPLC analysis; there was only a minor UV peak observed with tR = 5-6 min. (Fig. 2). The major UV peak eluted at tR = 23 min, as predicted from experiments with unlabeled methcathinone. When the HPLC fractions were analyzed for tritium, only one radioactive peak was observed and this peak coeluted with the methcathinone UV peak (Fig. 2). Overall radiochemical yield was 3.8% based on [3H]methyl iodide. The slight offset between the UV peak and the tritium peak is accounted for by the dead-space between the UV detector and the fraction collector.
Conclusion
In this paper, we describe the first synthesis of radiolabeled methcathinone. This psychostimulant drug is a potent inhibitor of plasma membrane catecholamine reuptake transporters with modest potency at the serotonin transporter. The availability of [3H]methcathinone will make new studies of methcathinone pharmacology possible. For example, it is possible to test whether methcathinone, like cocaine, is a pure inhibitor of monoamine transporters, or whether, on the other hand, it is also a transporter substrate and is accumulated inside cells. We have recently begun such studies in our laboratory. [3H]Methcathinone can also be used to identify high-affinity binding sites for amphetamine-like molecules in various tissues. Finally, this labeled compound should be useful in pharmacokinetic and metabolism studies to examine the absorption, distribution, and fate of methcathinone in vivo.