
#143 PROPYNYL
3,5-DIMETHOXY-4-(2-PROPYNYLOXY)PHENETHYLAMINE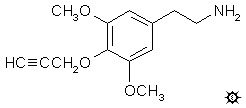
|
| [3D .mol structure] |
A suspension of 2.8 g LAH in 70 mL anhydrous THF was cooled to 0 °C with good stirring under He, and treated with 2.0 g 100% H2SO4. To this, a solution of 4.2 g 3,5-dimethoxy-4-(2-propynyloxy)phenylacetonitrile in 30 mL anhydrous THF was added very slowly. After the addition had been completed, the reaction mixture was held at reflux on the steam bath for 0.5 h, cooled to room temperature, treated with IPA to decompose the excess hydride, and finally with 15% NaOH to convert the solids to a white filterable mass. The solids were separated by filtration, the filter cake was washed with THF, and the filtrate and washes were pooled. After removal of the solvent, the residue was added to 100 mL dilute H2SO4, and washed with 3x75 mL CH2Cl2. The aqueous phase was made basic with dilute NaOH, and the product extracted with 2x75 mL CH2Cl2. After removal of the solvent under vacuum, the residue was distilled at 125-155 °C at 0.3 mm/Hg to provide 2.4 g of a light amber viscous liquid. This was dissolved in 10 mL IPA, acidified with concentrated HCl until a droplet produced a red color on dampened, external universal pH paper, and then diluted with 40 mL anhydrous Et2O with good stirring. After a short delay, 3,5-dimethoxy-4-(2-propynyloxy)phenethylamine hydrochloride (PROPYNYL) spontaneously crystallized. The product was removed by filtration, washed first with an IPA/Et2O mixture, and finally with Et2O. The yield was 3.0 g of white needles.
DOSAGE: 80 mg or more.
DURATION: 8 - 12 h.
QUALITATIVE COMMENTS: (with 55 mg) I have cold feet--literally--I don't mean that in the spiritual or adventurous sense. But also I am somewhat physically fuzzy. I feel that if I were in public my behavior would be such that someone would notice me. Everything was OK without any question at the ninth hour. I could walk abroad again.
(with 80 mg) There is a body load. The flow of people around me all day has demanded my attention, and when I had purposefully retreated to be by myself, there was no particular reward as to visuals or anything with eyes closed, either. Sleep was easy at midnight (the twelth hour of the experiment) but the morning was sluggish, and on recalling the day, I am not sure of the events that had taken place. Higher might be all right, but watch the status of the body. There certainly wasn't that much mental stuff.
EXTENSIONS AND COMMENTARY: No experiments have been performed that describe the action of this drug at full level. This compound does not seem to have the magic that would encourage exploration at higher levels.
| [ |
[Main Index] | [Forward |

